
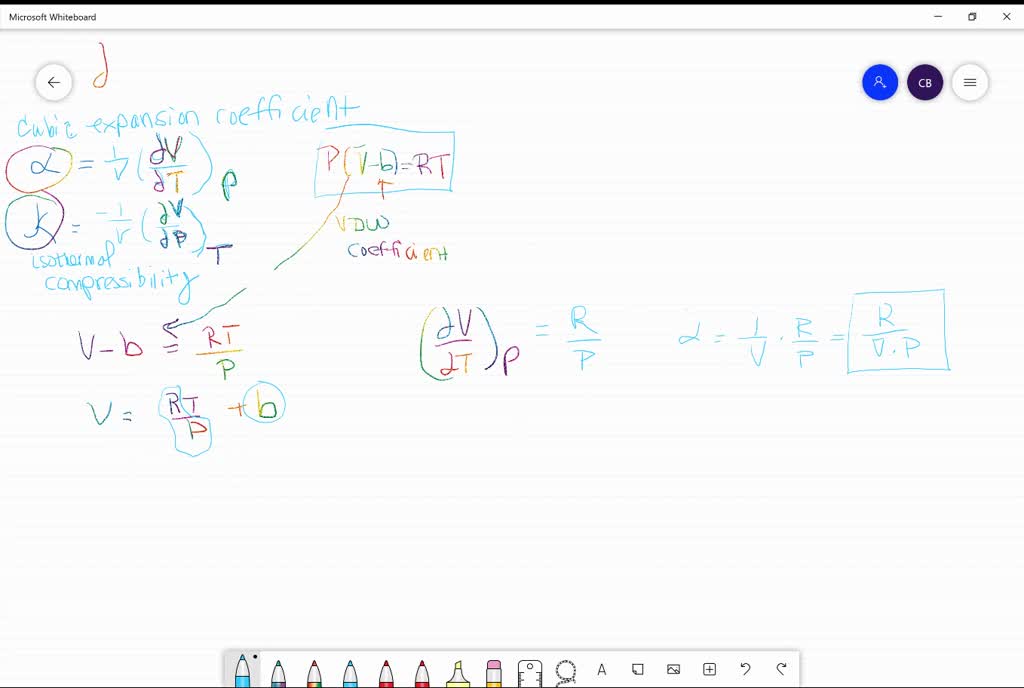
SOLVED:The isothermal compressibility κof a gas is defined in Problem 1.17, and its value for an ideal gas is shown to be 1 / P Use implicit differentiation of V with respect

SOLVED: Isothermal compressibility, Kr (kappa defined as: Derive the formula that - van der waals gas would have for kappa Show the unit analysis on your answer in part and check if





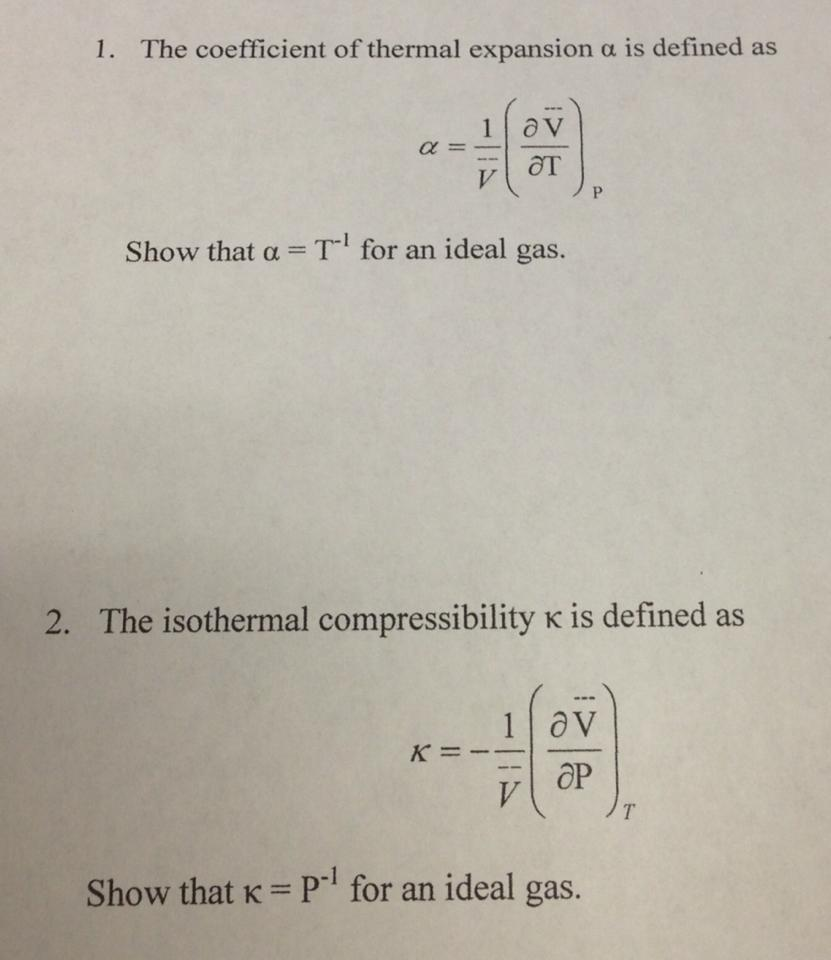



![Adiabatische Zustandsänderung: Erklärung und Darstellung · [mit Video] Adiabatische Zustandsänderung: Erklärung und Darstellung · [mit Video]](https://d3f6gjnauy613m.cloudfront.net/system/production/videos/001/164/61522b47ce21766a61f2668057056d9888c8fbf4/poster_Adiabatische_Zustands%C3%A4nderung.png?1679573945)



![Isentrope Zustandsänderung · einfache Erklärung · [mit Video] Isentrope Zustandsänderung · einfache Erklärung · [mit Video]](https://d3f6gjnauy613m.cloudfront.net/system/production/videos/001/479/0af0962f3bf48436d44794020e5410bfe23c50ab/Isentrope_Zustands%C3%A4nderung.jpg?1683199456)



